|
2/17/2024 0 Comments Fe iron charge
The most common siderophores fall into three main categories: hydroxamates, catecholates, and carboxylates. 
Siderophores are an incredibly diverse group of biocompounds, with hundreds of different types categorized. 5 Siderophores have the highest specificity and affinity for Fe(III), but are capable of forming complexes with other metallic ions, like Fe 2+, Zn 2+, Ga 3+, and Cr 3+. Siderophores are low molecular weight compounds that chelate iron and transport it into cells under low iron environments. 3, 4 To circumvent the lacking bioavailability of iron, these organisms synthesize and secrete siderophores. The insolubility means there is a low Fe(III) concentration (10 -18 M) of available iron too low to provide sustenance to microorganisms, which require a minimum concentration of 10 -8 M to live and grow. This is due to the aerobic nature of the environment, which causes iron to be present in the highly insoluble form, Fe(OH) 3. 2 This mechanism is more difficult, despite the prominence of iron in the atmosphere. The simpler of the two, is iron diffusion across cellular membranes. 1 There are two main mechanisms by which cells acquire iron. 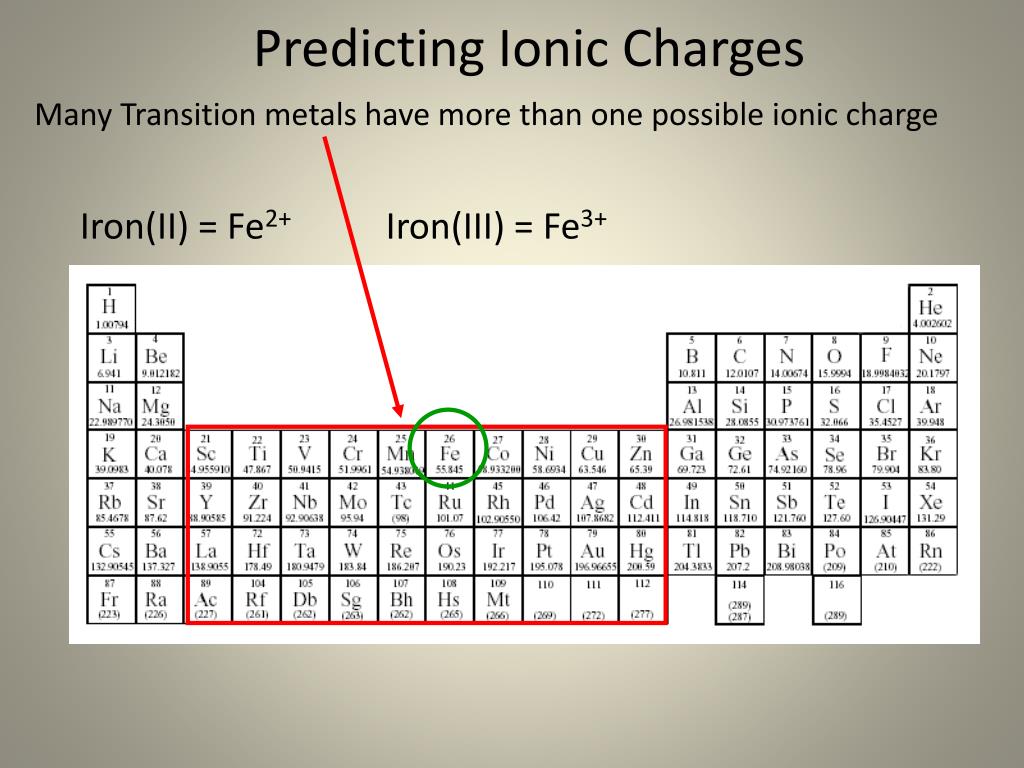
Animals receive this nutrient from diet, but plants and single-celled organisms must utilize different strategies. Most all living organisms require iron for crucial processes that allow for the life and growth of cells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
